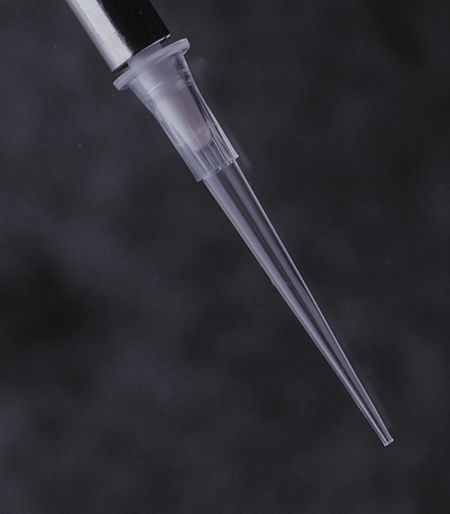
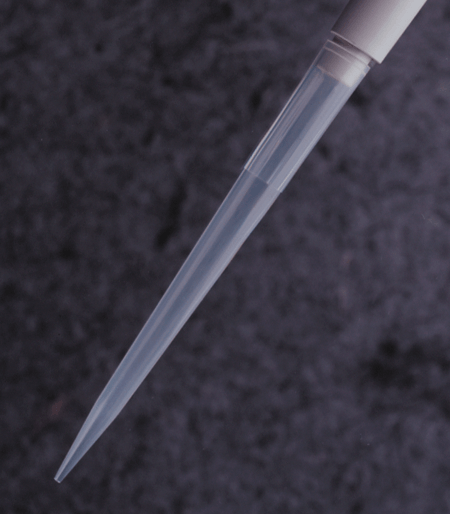
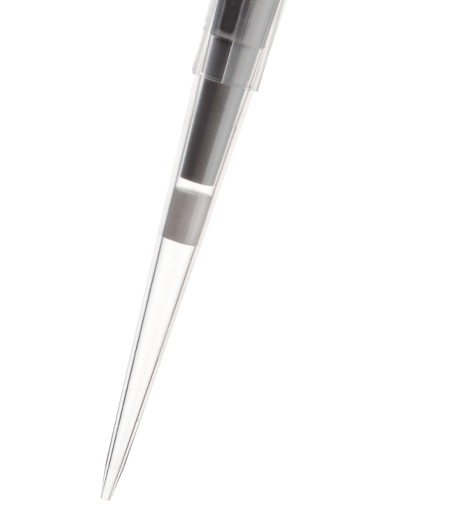
FlexTop™ Pipette Tips
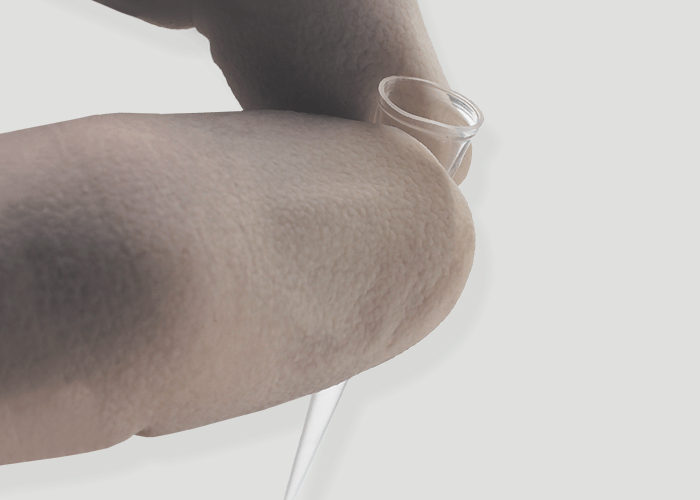
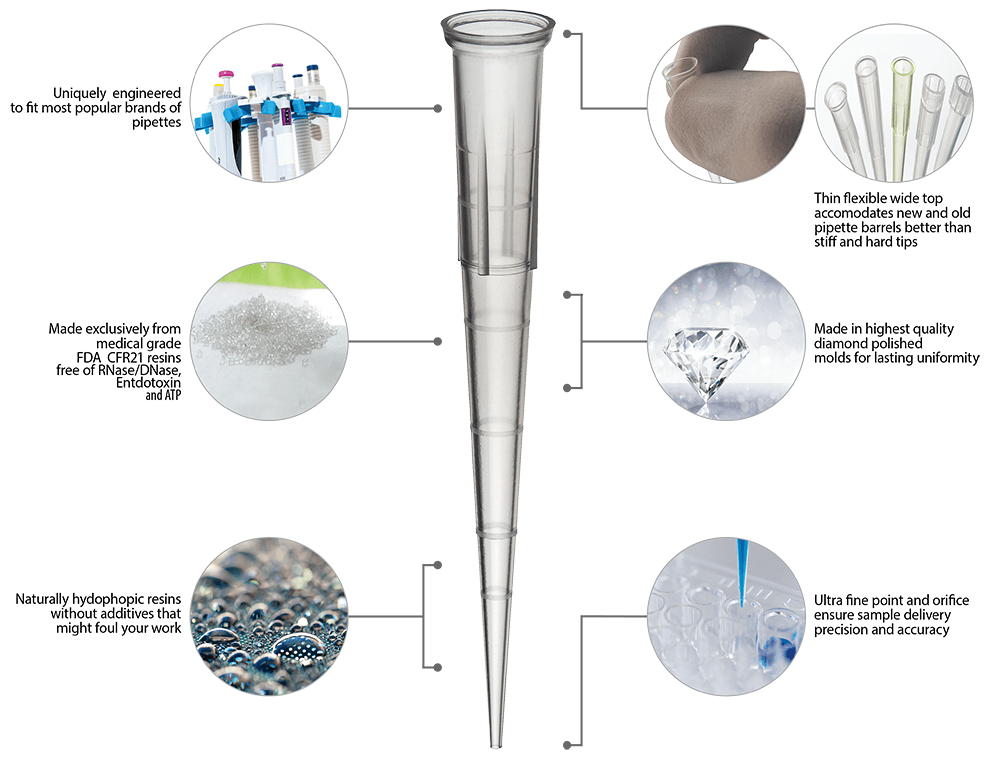
The repetitive work of mounting and ejecting pipette tips can place significant demands on your body and place you more at risk for some types of repetitive strain injuries. FlexTop™ Low Force pipette tips are designed to fit onto most popular pipettes and eject with less than 3Kg of force. This force is shown to help reduce the risk of strain injury. By using these Low Force tips you don’t have to invest in special pipettes to gain all the advantages of low force pipetting.

Higher Value
All our FlexTop™ tips in racks are packed in our trademarked Teal or Purple racks. These racks are made of polypropylene and are autoclavable repeatedly. They have sturdy latch hinges that won't come apart like lesser quality tip racks and a special snap lock designed so that it will not catch on your gloves. You can also get FlexTop™ tips in resealable zip style bags, in Eclipse™ refills, and in racks that have been sterilized using a validated 10-6 process.
See Eclipse RefillsThese ultimate tips feature a uniquely thin and flexible top that conforms to the pipette barrel unlike any other tips. They also include a non-beveled point that is so precise you can easily expel the tiniest amount of sample. And the top mounting is beveled to ensure that multichannel pipettes are easy to mount. FlexTop™ tips are made of clear 100% virgin polypropylene. Like all Labcon tips these tips are autoclavable along with their racks.
| Endotoxin Free (Non pyrogenic) Product samples are exposed to endotoxin-free water and the resulting extraction fluid is tested for contamination using the kinetic turbidimetric Limulus Amoebocyte Lysate (LAL) assay protocol and USP guidelines. All products tested must display less than 0.05 EU/ml to be certified free of endotoxin. |
|
| Nuclease Free (RNase/DNase) Product samples are exposed to nuclease-free water and the resulting extraction fluid is tested for nuclease activity on commercially available 7.5 kb Poly(A) tailed RNA (1µg) and HindIII-digested DNA (1µg) with a one hour 37°C incubation in appropriate buffers. Results are visualized on an agarose gel with appropriate positive and negative controls. Extraction fluid samples must show no degradation of the nucleic acids by the extraction fluid has occurred for the product to be certified as RNase-free and DNase-free |
|
| Adenosine Triphosphate (ATP) Product sample surfaces are tested for the presence of adenosine triphosphate (ATP) using a controlled bioluminescence reaction to detect contamination. Luminescence data is compared to results generated by ATP-free surfaces and surfaces with known amounts of ATP as a positive control. The relative light units result must indicate less than 2 X 10-12 mg/µl of ATP for the product to be certified as ATP free. |
|
| Validated Sterilization Labcon products are sterilized by radiation sterilization within a dose range of 12-35 kGy. The dose range necessary for the stated sterility assurance level is continuously audited through quarterly bio-burden and sterility validation studies performed according to the ANSI/AAMI/ISO 11137 standard by an independent laboratory. This dosage is sufficient to guarantee a sterility assurance level of 10-6. |
| Bovine Spongiform Encephalopathy-Transmissible Spongiform Encephalopathy These products contain resins that are processed under conditions proven to exceed the European Union standard as listed in the 22nd Commission Directive EC 98/16/EC of March 5th, 1998 as annexed to council Directive EC 76/768/EEC and further Amendment 419 Annex II of 12 June 2001. |
|
| Medical Grade (USP) U.S Pharmacoepia Methods and Guidelines (U.S.P Class VI) are used if applicable. We only use medical grade resins and pre-test all resins for contaminants prior to use. Resins are compliant with FDA CFR title 21-177.1520, 178.3295, 178.3297. |
|
| California Prop 65 No Labcon manufactured disposables contain any of the “listed chemicals” as referenced in the California Safe Drinking Water and Toxic Enforcement Act of 1986, (Prop 65) as revised May 25, 2018. |
|
| Phthalates & Oleamide All our resins are medical grade and are certified free of Bisphenol A (BPA), Oleamide, DiHEMDA, and Phthalates. |
|
| Substances of Very High Concern & REACH All Labcon products are compliant with RoHS Directive 2002/95/EC/-2011/65-2015/863, are free of Substances of Very High Concern (SVHC), and are EU REACH Regulation (EC) No 1907/2006 compliant. |
|
| Origin These products are Made in USA and all components meet the requirements for US origin under the NAFTA agreement. |
| U.S. FDA Registered Labcon is a U.S. FDA registered medical device manufacturer. Our facility is registered by the U.S. government to comply with CFR21 GMP regulations for manufacturing medical devices. |
|
| CE Compliant As applicable Labcon products meet the requirements for CE marking under regulation 2017/746. |
|
| ISO 9001 Quality Registration Labcon has been registered to the ISO 9001 quality management system standar since 1997 and maintains registration to ISO 9001:2015. |
| Material | 100% Virgin Polypropylene |
| Grade | Medical |
| Rating | USP Class VI |
| Color | Natural (Clear) |
| Length | Various |
| Fits | Most popular pipette brands |
| Approved Use | Medical, Research, Industrial, Food & Beverage |
| Lot Expiration | 4 years |
| Sterile Expiration | 3 years |
96 Rack
These racks offer workhorse reliability and versatility. They have heavy hook hinges that are extremely durable and an ergonomic snap latch that won't catch gloves. Smooth rounded edges and reinforced seal make these racks durable additions to your lab. Strong enough to be autoclaved many times. The separate rack insert includes a beveled top and extra sturdy surface for multi-channel pipetting. A vent in the rack sides allows steam autoclaving to be more efficient and assures your tips are sterile.